QMS ISO 13485 ISO
ISO 13485:2016 is based on the ISO 9001 process model approach and is a management systems standard specifically developed for the manufacture of medical devices. ISO 13485 certification is especially relevant to manufacturers that wish to demonstrate applicable regulatory requirements and by organisations whose services support medical device manufacturers. These companies need to get certified to ISO 13485. ISO 13485 applies to organisations of all types and sizes, except where explicitly stated otherwise. ISO 13485 contains requirements that are essential for any organisation operating at any tier in the medical device and pharmaceutical supply chain, including design, production, installation, storage, development and service. Looking to implement an ISO 13485 management system? Not sure where to start? Take a look at our ISO 13485 Implementation Guide here. Statistics straight from the most recent ISO Survey. ISO 13485 has seen a 33.1% increase in worldwide certificates in 2020, showing the growth and importance of UKAS accredited certification in recent times. The most recent version of ISO 13485 is reviewed every five years and revised according to the new requirements and needs of the industry. As an internationally recognised standard of quality and safety for medical device manufacturing, having ISO 13485 certification helps companies get recognised as more reputable, trustworthy providers.
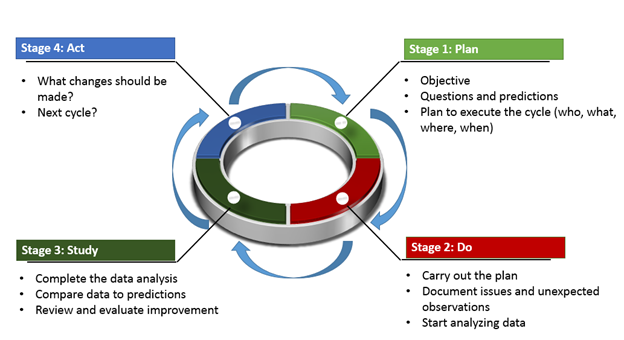
This standard allows companies to reduce safety and legal risks while creating more economical work environments.

The standard contains specific requirements for manufacture, installation and servicing of medical devices and calls for: Its primary objective is to facilitate harmonised medical device regulatory requirements.

ISO 13485 certification is a valuable credential put in place to keep professionals and customers safe in clinics, hospitals and other medical settings. ISO 13485 is the medical industry's optimal medical device standard, which ensures that all medical devices meet the proper regulatory compliance laws and customer needs.
